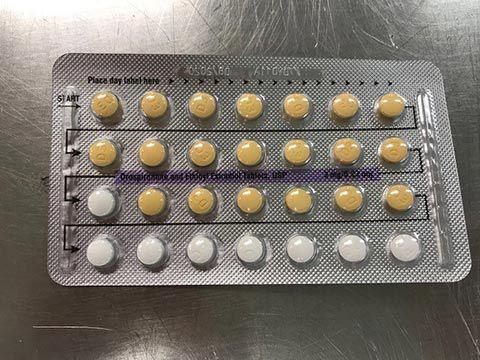
The FDA has announced that a pharmaceutical company out of Florida is voluntarily recalling some birth control pills because they may possibly contain defective blisters with incorrect tablet arrangements and/or an empty blister pocket.
Apotex Corp . is voluntarily recalling four lots of Drospirenone and Ethinyl Estradiol Tablets, USP to the patient /user level.
As a result, users may not take a tablet due to a missing tablet or take a placebo instead of an active tablet, causing an adverse event or even pregnancy.
No case of unintended pregnancy has yet been reported, according to Apotex.
Users with questions regarding this recall are urged to contact their pharmacy and healthcare provider for medical advice and return the impacted packages to their pharmacist.
The following lots have been recalled: 7DY008A, 7DY009A, 7DY010A, and 7DY011A. The outer carton displays NDC# 60505-4183-3 and contains three inner cartons (NDC# 60505-4183-1). The expiration date is listed as 8/2020.
For more information click here



